Unlocking safe and reliable access to a wider range of emotional and cognitive states

About
What we do
Mindstate Design Labs is a clinical-stage AI neuroengineering platform and drug development company addressing the mental health crisis by creating a new way that we can change our minds.

Osmanthus
Our AI-powered platform maps the biological basis of emotion, perception, and cognition. Synthesizing 60,000 human reports of psychoactive drug effects with biochemical data, Osmanthus identifies how specific neurotransmitter receptor combinations produce distinct emotional and cognitive states.
Osmanthus applies multi-agent large language models, frontier & custom embedding models, Bayesian neural networks, and other statistical & machine learning methods to connect the leading datasets of psychoactive phenomenology and pharmacology.
Our Pipeline
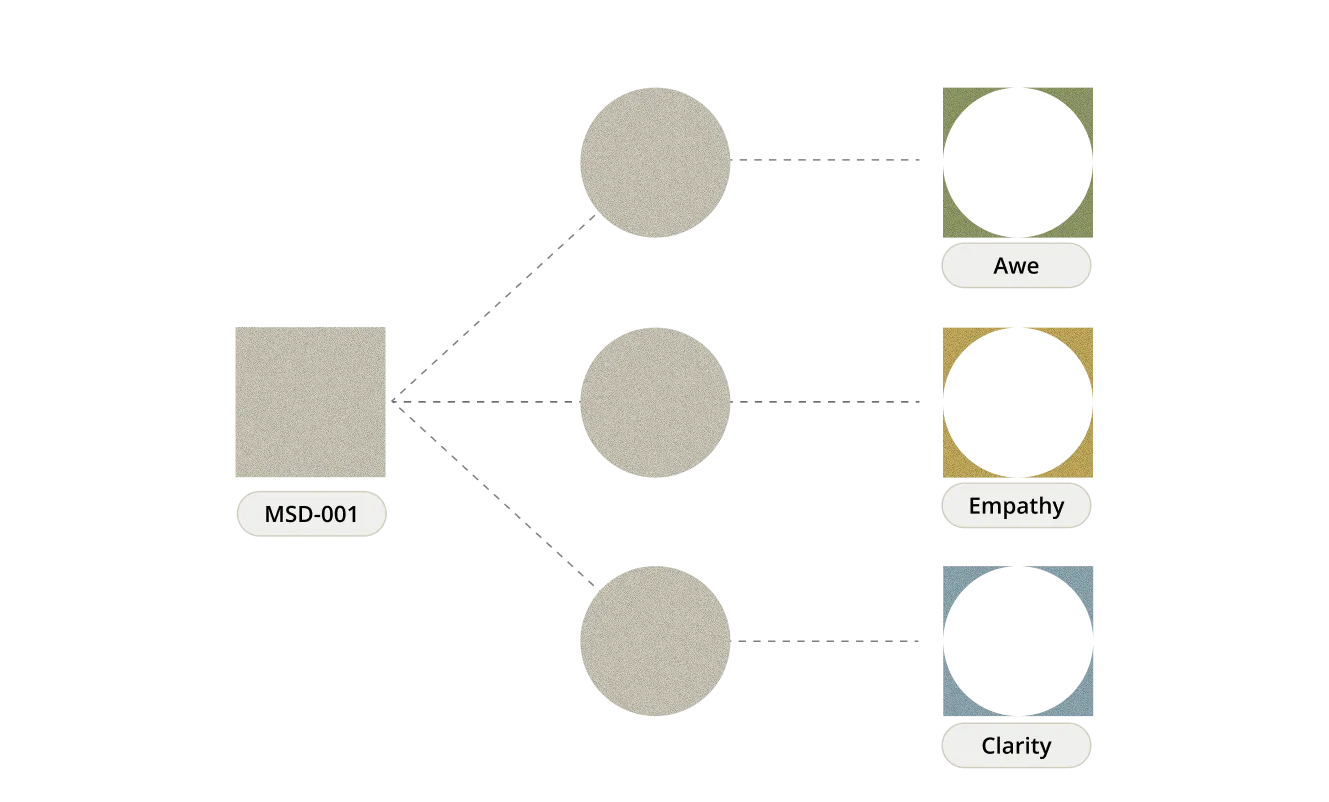
Lead Asset: MSD-001
MSD-001 is a pharmaceutical compound that can temporarily enable significant emotional & cognitive flexibility.
We’re
developing multiple combinations with the base ingredient of MSD-001.
Each combination is built to reliably induce a state like empathy, awe,
or enhanced aesthetic perception - offering new therapeutic
possibilities to transform our deepest psychological afflictions.
Our Pipeline

MSD-001 is currently in human studies to demonstrate that profoundly therapeutic emotional and cognitive states can be precision-designed.
MSD-X02 series is designed to act on a novel central nervous system target to produce select psychoactive effects.
Future programs based on high-throughput Osmanthus screening optimize for single-molecule, multi-target action producing a broader range of psychoactive effects.

Our Team

Dillan DiNardo
Dillan
DiNardo is a biotechnology investor and operator with a background in
venture capital. Prior to Mindstate Design Labs, he worked in a $1
billion venture fund, executing and managing more than fifty biopharma,
medical device, and health tech investments ranging from pre-seed to
publicly traded companies. In addition to his investing capacity, he
took an operating role in over a dozen early-stage spinout companies.
His experience spans a variety of disease indications and modalities,
with a focus on deep-tech applications such as cell and gene therapies.
Prior to his venture roles, he worked in finance for large international
accounting and consulting firms as a transaction-focused consultant,
covering the full investment life cycle from seed-stage startup funding
to Fortune 500 mergers, acquisitions, and divestitures. Dillan holds an
MBA and a Bachelor's in Finance from Robert Morris University.


Thomas Ray, PhD
Thomas Ray, Scientific Founder: Dr. Ray is an evolutionary biologist, iconoclast, and polymath who has made seminal contributions to multiple fields of science. His paradigm-shifting work has been featured in dozens of media outlets, including Time Magazine, The New York Times, and Wired. Enrolling in university at the age of 16, as an undergraduate he was already publishing in the journal Science about new phenomena of nature he discovered with the natural history method. He brought the same natural history approach to the field of artificial life; with no training in computer science, he designed a new CPU architecture, a new operating system, and a new machine language. He created the first known instance of evolution by natural selection apart from life on earth - the first true artificial life program. For the past two decades, he has devoted his research to the natural history study of psychoactivity, synthesizing human experience data and biochemical data to develop hypotheses concerning the biological basis of the diverse varieties of psychoactive drug effects. Dr. Ray holds a PhD in Biology from Harvard University.


Rob Silva, PhD
Dr. Silva is an accomplished neuroscientist and drug development executive with over 20 years of experience in research and pharmaceutical development leading global drug development teams to advance treatments in psychiatric and neurological indications from preclinical through Phase III. He has led several successful development programs resulting in multiple global marketing approvals for CNS products LATUDA® and FINTEMPLA® across schizophrenia, bipolar disorder, Lennox-Gastaut Syndrome, and Dravet Syndrome. In addition to his prior role as Global Head of Development at Mind Medicine, he has held executive and director-level leadership positions at Noema Pharma, UCB, Ipsen Bioscience, and Sunovion Pharmaceuticals. Dr. Silva began his drug development career as a Clinical Scientist at Merck after completing his post-doctoral fellowship at Columbia University College of Physicians and Surgeons. He holds a PhD in Neuroscience from City University of New York.


Tilmann Brotz, PhD
Dr.
Brotz is a neuroscientist and drug development expert with over 20
years of experience leading toxicology, DMPK, in-vivo pharmacology,
clinical pharmacology and diagnostic groups. Most recently Dr. Brotz was
Sr. Director, Development Sciences at Achaogen where he contributed to
multiple programs from lead optimization through Phase 3. Prior to
Achaogen, Dr. Brotz worked as head of preclinical development and as a
consultant for a number of emerging companies. He is an advisor for the
CLSI FAST Program and a member of the Scientific Review Board of the
Alzheimer’s Drug Discovery Foundation. Dr. Brotz holds a PhD in
Neuroscience from the University of Tübingen.


Ken Colley, MD, PhD
Dr. Colley is a physician-scientist with more than 20 years of experience focused on developing small and large molecule therapeutic products from R&D through human proof-of-concept mid-stage clinical studies. As Executive Medical Director, Medical Monitor, and Head of Product Development for emerging biotech and large biopharma companies, he has overseen development programs across clinical pharmacology and translational medicine, first-in-human, Phase 1, and human proof of concept Phase 2+ studies. In addition to completing numerous successful Phase 1 and Phase 2 studies, Dr. Colley’s experience also extends into later stage medical safety analyses, submission dossiers, market approval, and medical affairs.
Dr. Colley holds an MD and PhD in Molecular Pharmacology from Georgetown University and a Bachelor’s in Biological Sciences from Stanford University.


Jianhong Wang, PhD
Dr. Wang has over 20 years of experience contributing to the discovery of multiple clinical candidates, leading cross-functional discovery teams, and serving as a clinical pharmacology subject matter expert. Previously Dr. Wang was Executive Director of Clinical Pharmacology and DMPK at Cortexyme. Earlier in her career as a DMPK Research Scientist at Gilead Sciences, she led the nonclinical team in completing the nonclinical IND and NDA package for Biktarvy®, which is the best-selling medicine to treat HIV. Dr. Wang holds a PhD in Analytical Chemistry from Iowa State University.


Jessica Smith Beaver, PhD, MLS
Dr. Beaver is a seasoned regulatory leader with a strong track record in regulatory affairs, healthcare compliance, quality assurance, and risk management. Her experience includes small molecule and biologics development for drug candidates across discovery, early and late-stage development, and marketed products. Beginning her career at Johnson & Johnson as a Clinical Pharmacokinetics Leader, Dr. Beaver subsequently held senior regulatory roles at Targacept, Kowa Pharmaceuticals, and Verinetics. She holds a PhD in Pharmacokinetics & Drug Metabolism from the University of North Carolina at Chapel Hill and a Master of Legal Studies in Business & Compliance Law from Washington University in St. Louis.


Tom Richardson, PhD
Dr. Richardson has over 20 years of medicinal chemistry experience in integrated drug discovery, developing and implementing research strategies from lead generation to pre-IND candidate selection. He previously served as Discovery Program Leader at Cavion, Inc. (acquired by Jazz Pharmaceuticals), developing novel T-Type calcium channel inhibitors for neurological and oncology indications. He is the co-inventor of 15 patents and co-author of several publications in peer-reviewed journals, and has a broad knowledge base and experience in crafting new intellectual property to secure patent space. Dr. Richardson holds a PhD in Organic Chemistry from The Ohio State University.


Antonio Socorro
Antonio Socorro has over 20 years of international experience in CMC, CQV, quality assurance, technical operations, and supply chain. His cross-functional product development roles have included both commercial and clinical manufacturing of small molecule and biologic products with companies including Merus N.V, Glycosystem Therapeutics, and EUSA Pharma, where he established global CDMO partner networks. His earlier roles included technical leadership in biologics manufacturing at Ipsen and Custom Pharma Services. Antonio holds a Bachelor’s in Molecular Genetics from Curtin University.


Tegan Johnson
Tegan Johnson has built on her background as an engineering physicist designing particle accelerators to move into project and program management for complex medtech development. Most recently, she served as Director of Program Management at Openwater, a neurodiagnostic medical imaging company where she led the inaugural regulatory program and drove the development program from early hardware proof-of-concept to its first human studies. Prior to her role at Openwater, she served as Project Manager at Varian Medical Systems where she led product design development, risk hazard analysis, verification & validation, and compliance testing to FDA standards for a number of projects, each with teams numbering between 5-20 personnel. Tegan holds a Bachelor's in Psychology and Physics from Otterbein University and a Master's in Particle Beam Physics from Indiana University in conjunction with the Stanford Linear Accelerator Center.


Michael Hoyos
Michael Hoyos is an investor and operator with a focus on frontier therapeutics and a background in venture capital, private equity, and consulting. He previously worked at McKinsey & Company, where he led engagements for Fortune 500 companies and nonprofit clients across the firm’s life sciences, healthcare, and social sector practices.
Before McKinsey, Michael co-founded an early-stage venture firm investing in novel CNS therapeutics to address mental health, addiction, and pain. He also led an advisory firm supporting ultra-high-net-worth families in managing direct private equity portfolios exceeding $100 million. He began his career at JPMorgan.
Michael holds an MBA from the University of Chicago Booth School of Business and a BS in Economics from the Wharton School at the University of Pennsylvania.


Josie Kins
Josie Kins is a pioneer and prominent leader in the online psychonautic community, having spent the last 12 years formalizing the documentation of subjective psychedelic effects. She spearheads the effort to develop a detailed index of the diverse varieties of the psychedelic experience, which will enable new psychometric scales and computational efforts to map the biological basis of subjective psychedelic experience.
Josie brings a deep familiarity with psychedelic phenomenology and a unique ability to precisely articulate the range of psychedelic experience. As the founder of PsychonautWiki.org, Josie grew the site into a leading source of information about the phenomenology of psychedelic drugs with over one million unique monthly visitors.
She is also the founder of EffectIndex.com which features a granular taxonomy of the subjective psychedelic experience. Recognizing that verbal descriptions of psychedelic phenomenology would greatly benefit from complementary visual and auditory representations, she also founded the r/replications subreddit which has over 140,000 subscribers and has launched the careers of multiple visionary artists.


Drew Thieme, PhD
Dr. Thieme is a cheminformatician with deep expertise in the areas of data science, biochemical and subjective experiential data curation, computational chemistry, medicinal chemistry, psychopharmacology, pathophysiology, and drug discovery and development. He employs machine learning techniques, quantitative structure-activity relationship modeling, molecular modeling, and biomedical knowledge graph analysis to develop virtual screening platforms oriented toward the identification of novel candidate molecules for targeted applications.
Dr. Thieme's dissertation focused on harmonizing raw subjective scent data via natural language processing, predicting subjective scent perception profiles from chemical structure of odorants, and leveraging biomedical knowledge graphs to draw connections between perceptual processes and various disease states. Additionally, Dr. Thieme has worked to formally define the concept of Clinical Outcomes Pathways, a new paradigm for the classification, characterization, and comparison of drug action. Now, at Mindstate Design Labs, Dr. Thieme is adapting these methods to the domain of subjective psychedelic experience to drive discovery of novel psychedelic drugs and precision design of modified conscious states.
Dr. Thieme holds a PhD in Pharmaceutical Sciences from the University of North Carolina at Chapel Hill and a Bachelor's Degree in Pharmaceutical Sciences from Purdue University.


Ahnjili ZhuParris, PhD
Ahnjili ZhuParris is a data scientist, machine learning engineer, and cognitive neuroscientist specializing in applying machine learning techniques to generate insights from complex heterogeneous data. She was previously a data scientist at the Centre for Human Drug Research (CHDR), a leading clinical research organization, where she developed a data science library for analyzing clinical trial data.
While at CHDR, Ahnjili was also a PhD Candidate at Leiden University Medical Center and Leiden Institute of Advanced Computer Science, where her thesis focused on translating self-reported outcomes, physiological, and behavioral data into meaningful clinical insights. She created new biomarkers that enabled passive collection of informative health data during clinical trials and used those biomarkers to develop machine learning models that classified clinical diagnosis, estimated symptom severity, and predicted future clinical states.
Ahnjili’s prior research has also explored the impacts of LSD, methylphenidate, and mindfulness on cognitive flexibility. In addition to her academic pursuits, she hosts workshops and installations that aim to demystify the mechanisms, biases, applications, and potential challenges associated with AI algorithms.
Ahnjili holds a Bachelor's Degree in Neuroscience from Edinburgh University and a Master's Degree in Cognitive Neuroscience from Radboud University. She holds a PhD in Advanced Data Analytics from Leiden University.

Our Advisors

Frederick Barrett, PhD
Dr. Barrett is the Associate Director of the Johns Hopkins Center for Psychedelic and Consciousness Research, the first academic psychedelic research center in the U.S. and the largest in the world. Dr. Barrett was the PI for the first federally funded research since the 1970s administering a classic psychedelic to people with psychedelic effects as the primary focus. He is now leading a number of ongoing studies aimed at better understanding the psychological, biological, and neural mechanisms underlying therapeutic efficacy of psychedelic drugs.


Danilo Bzdok, MD, PhD
Dr. Bzdok is a medical doctor and computer scientist with a dual background in systems neuroscience and machine learning algorithms. He currently serves as Associate Professor at McGill's Faculty of Medicine and as Canada CIFAR AI Chair at Mila - Quebec Artificial Intelligence Institute, including cross-appointments at the McConnell Brain Imaging Center, Montreal Neurological Institute, Ludmer Centre for Neuroinformatics and Mental Health, and the School of Computer Science at McGill University. Dr. Bzdok's interdisciplinary research activity centers on narrowing knowledge gaps in the brain basis of human-defining types of thinking, with a special focus on the higher association cortex in health and disease.
After completing his medical training at RWTH Aachen University, Université de Lausanne, and Harvard Medical School, Dr. Bzdok completed one PhD in cognitive neuroscience at Research Center Juelich and one PhD in computer science in machine learning statistics at INRIA Saclay and Neurospin.


Robin Carhart-Harris, PhD
Robin Carhart-Harris is the founder of the Centre for Psychedelic Research at Imperial College London, the world's first formal academic center for psychedelic research. He is now the Director of the Neuroscape Psychedelics Division and the Ralph Metzner Distinguished Professor of Neurology and Psychiatry at the University of California San Francisco. Dr. Carhart-Harris has been one of the foremost leaders of the psychedelic renaissance, having authored well over 100 psychedelic research publications with more than 15,000 collective citations. He has designed a number of functional brain imaging studies with psilocybin, LSD, MDMA, and DMT, and has completed multiple clinical trials of psilocybin for depression.


David Erritzoe, MD, PhD
David Erritzoe is the Deputy Head and Clinical Director of the Centre for Psychedelic Research at Imperial College London and Director of the CNWL-Imperial Psychopharmacology & Psychedelic Research (CIPPRes) Clinic at St. Charles Hospital. With more than 70 research publications and deep expertise in psychopharmacological research using brain imaging techniques, he is currently heading investigations on several psychedelic compounds including investigations on the mechanisms of ketamine's therapeutic effects, the role of 5HT2A receptors and 5HT release capacity in depression, and Phase I & II clinical trials on DMT in major depression.



Rand Hindi, PhD
Dr.
Hindi is an entrepreneur and deeptech investor. He is the CEO at Zama,
an open source homomorphic encryption company, and an investor in more
than 30 companies across privacy, AI, blockchain, medtech, and
psychedelics. Dr. Hindi started coding at the age of 10, founding a
social network at 14 and a web agency at 15 before getting into machine
learning at 18 and starting a PhD at 21. He then created Snips, an AI
startup that was acquired by Sonos and is now powering the voice
assistant in over 20 million devices.
Dr. Hindi has been
elected as a TR35 by the MIT Technology Review and as a "30 under 30" by
Forbes, was a lecturer at Sciences Po in Paris, and is an advisor to
multiple companies. He was previously a member of the French Digital
Council where he focused on AI and Privacy issues. Dr. Hindi holds a PhD
in Bioinformatics from University College London as well as two
graduate degrees from Singularity University in Silicon Valley and THNK
in Amsterdam.


Andrew Newberg, MD
Dr. Andrew Newberg is a neuroscientist who studies the relationship between brain function and various mental states with a focus on altered states of consciousness outside those produced by existing psychedelic compounds. He is a pioneer in the neuroimaging and neurological study of transpersonal experiences, which are highly correlated with positive enduring treatment outcomes in psychedelic therapy. Dr. Newberg is the author of The Varieties of Spiritual Experience and a number of other books largely focused on how brain function is associated with various mental states - in particular, religious and mystical experiences.


Peter Wipf, PhD
Dr. Wipf is a Distinguished University Professor of Chemistry at the University of Pittsburgh. His research centers on the discovery of new pharmaceuticals including the total synthesis of natural products, organometallic and heterocyclic chemistry, and medicinal chemistry. Dr. Wipf is the author of more than 500 academic publications with over 37,000 citations, and he serves on a number of advisory and editorial boards in the fields of chemistry and pharmaceuticals.

News
Follow our journey as we repurpose the mechanisms of psychedelics to pioneer a new paradigm in mental health therapeutics.
Press Releases
Check out the latest news

Inspired to make an impact?
We're always on the lookout for exceptional talent. Submit an application below, and we'll reach out when an opportunity aligns with your skills and aspirations.
Apply Now